WHO Calls US-Funded Guinea-Bissau Hepatitis B Trial Unethical
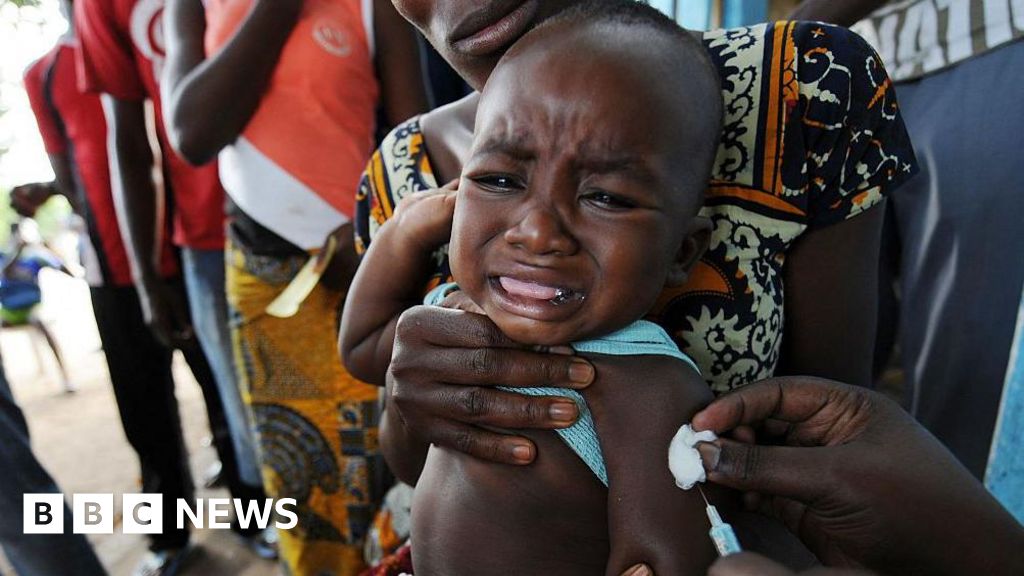
WHO said the US-funded study would withhold a proven hepatitis B birth-dose from newborns in Guinea-Bissau, affecting 14,000 babies and funded with $1.6 million.
Overview
On February 13, 2026, the World Health Organization said a planned US-funded hepatitis B newborn trial in Guinea-Bissau was "unethical".
The study, funded by a $1.6 million CDC award published in the Federal Register in December, planned to randomize 14,000 newborns to receive hepatitis B vaccine at birth or at six weeks.
The Guinea-Bissau government suspended the project last month amid public outrage, and critics including former health minister Magda Robalo said citizens were "not guinea pigs".
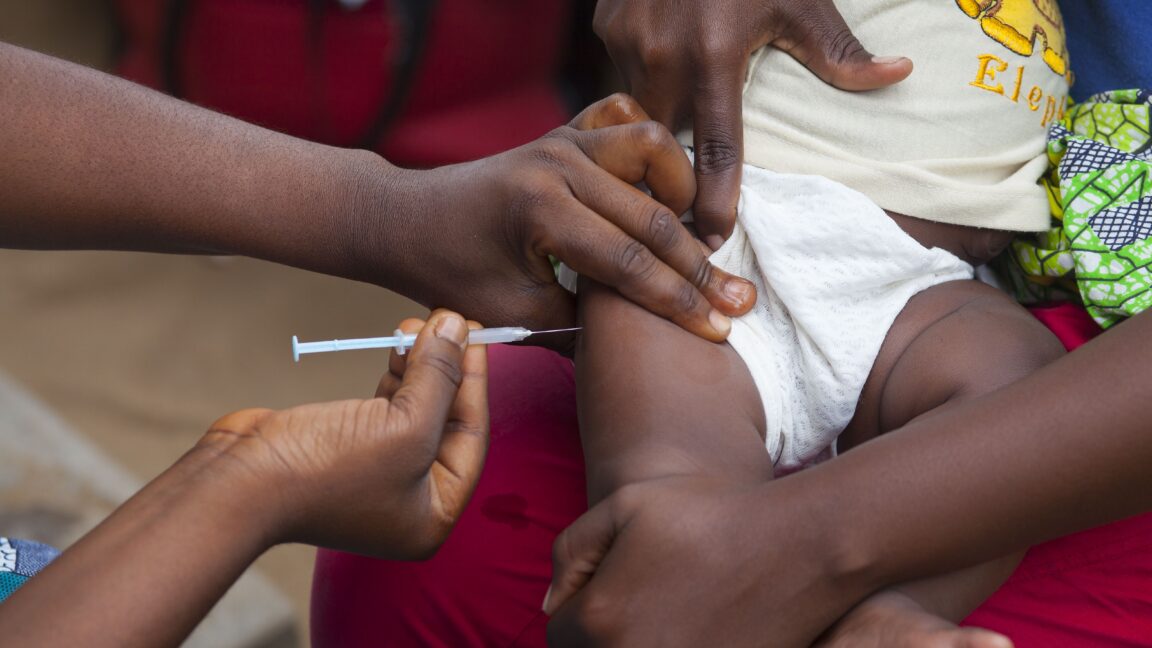
WHO said the birth-dose vaccine prevents mother-to-baby transmission in 70–95% of cases and that more than 12% of Guinea-Bissau's adult population has chronic hepatitis B.
WHO said it would help accelerate Guinea-Bissau's plan to introduce a birth dose nationwide by 2028 while the trial remains suspended and broader policy debates continue.
Analysis
Center-leaning sources frame the story as ethically alarming and politically driven, foregrounding WHO's 'unethical' finding, CDC funding and an 'anti-vaccine' health secretary, and questioning researchers' credibility. Through loaded verbs ('slams', 'blasting'), selective emphasis on condemning experts, and highlighting procedural irregularities, coverage steers readers toward viewing the trial as harmful and politicized.
FAQ
WHO cited significant concerns about the trial's scientific justification, ethical safeguards, and alignment with research principles, as it would withhold the proven birth-dose vaccine, which prevents 70-95% of mother-to-child transmissions, without ensuring minimum harm reduction like screening pregnant women.
The Guinea-Bissau government has suspended the trial pending further technical reviews by its National Medicine Regulatory Authority and National Ethical Committee.
The trial planned to randomize about 14,000 newborns, with half receiving hepatitis B vaccine at birth and half at six weeks, funded by a $1.6 million CDC award, led by the Bandim Health Project to assess overall mortality, hospitalizations, and other outcomes.
Over 12% of adults have chronic hepatitis B, and infection in children under five is about 2%, above global targets. Guinea-Bissau currently vaccinates at 6 weeks but plans nationwide birth-dose introduction by 2028.
The trial is led by Denmark's Bandim Health Project (SDU), funded by US CDC, Pershing Square, and Bluebell Foundations. Critics include WHO, former Guinea-Bissau health minister Magda Robalo, and Africa CDC supports the suspension decision.